Quick Summary of Energy & the Living Cell
The Cell Membrane
The function of the cell membrane controls what enters and leaves the cell. It allows passage of water, holds the contents of the cell together, and allows for transmembrane communication. There are cell markers that identify all types and identify all cells as your own. The membrane is made up of lipids and proteins. Lipids include fat, phospholipids, glycolipids, and steroids. Fats are composed of a glycerol backbone with a carboxyl functional group. Phospholipids are a major component of all cell membranes. They are made of a glycerol backbone, two fatty acid chains, and phosphorylated alcohol. Within membranes, the phospholipids orient themselves so as to form a bilipid layer where the polar heads are in contact with water and form H bonds and the non-polar tails are away from water.
The membrane is made up of amino acids. There are 20 different amino acids grouped into five categories. Each of the amino acids have different properties. Some are acidic, some are basic, some a hydrophobic, and some are hydrophilic. Since proteins are long chains of amino acids, depending on the amino acid sequence, different parts of a protein will have very different properties.
The membrane contains proteins and lipids in varying amounts depending on the type of membrane. Proteins are 1.5 to 4 times greater than phospholipids by weight. There are usually a higher amount of phospholipids than proteins in the cell membrane. Proteins can be attached loosely to the outside of the membrane, or they can actually span the membrane.
Energy
In biological systems, energy is obtained from chemical bonds. When bonds are broken, energy is required. When bonds are formed, energy is released. Each type of bond has a specific bond energy. It can be predicted whether a chemical reaction will release or require heat by using bond energies. When there is more energy used to form the bonds than to break the bonds, heat is given off. This is known as an exothermic reaction. When a reaction requires an input of energy, it is known as an endothermic reaction. Activation energy is the ability to break bonds.
ATP
ATP is the short form for Adenine Triphosphate. It is the universal energy currency of the cell. ATP is required to produce large molecules for growth and repair, it provides activation energy for reactions in cells, it is used in muscle movement, for active transport, for heat, and for electrical signals. In ATP there are three subunits. The first is ribose, which is a five carbon sugar. The second is adenine, which is two rings of carbon and nitrogen. The third subunit is t, which is three covalently bonded phosphate groups. The useful energy of the ATP molecule is contained in the bond called a phosphorhydride bond.
With the help of enzymes, the activation energies of all the reactions in the cell are less that 7.3 kcal/mol. Therefore, ATP releases enough energy to make reactions proceed.
Enzymes
Enzymes are a protein molecule made of many amino acids. They fold into a 3D shape which is determined by the properties of the amino acids. There are weak hydrogen bonds that keep the 3D structure. The hydrogen bonds are easily disrupted by heat or pH. The 3D structure is very important to protein function. If the structure is disrupted, the enzyme is less useful. The 3D structure produces active sites for molecules. This is where chemical reactions occur. These chemical reactions are very specific.
The action of enzymes is to speed up reactions. Firstly, a substrate fits into enzyme active site. The enzyme then changes shape to ensure a tight fit. Amino acids on an enzyme are in close contact in bonds of a substrate. The enzyme's amino acids stress or distort the bond so it is easier to break. Products are then released and the enzyme can be reused.
Enzymes are have many different properties. They increase the rate of reaction without being consumed, and their presence doesn't alter the products. Activity is affected by pH and temperature, and varies with amount of substrate and enzyme. Reactions are reversible, and the enzymes are very specific for certain reactions. Enzyme activity must be regulated very carefully. It is enzymes that determine which reactions will occur when.
Coenzymes are not proteins. They can be both organic and inorganic. They can perform a number of roles. They attract substrate to the binding site, help reactions proceed after binding, and accept or donate electrons.
Cellular Respiration
The functions for respiration are for energy production, and for the formation of building blocks for larger molecules. Cellular respiration takes four steps:
Glycolysis
Glycolysis is the most primitive part of cellular respiration. It is an anaerobic process, which doesn't require oxygen. Each reaction is catalyzed by a different enzyme. It occurs in the cytoplasm. The end product is pyruvate. The steps of glycolysis are as follows:
-
Glucose
 Glucose-6-phosphate, This is a phosphorylation reaction because a
phosphate is gained. This is also an endothermic reaction which
requires energy from the exothermic reaction ATP
Glucose-6-phosphate, This is a phosphorylation reaction because a
phosphate is gained. This is also an endothermic reaction which
requires energy from the exothermic reaction ATP  ADP + Pi
ADP + Pi -
Glucose-6-Phosphate
 Fructose-6-phosphate, This is simply a rearrangement called an isomerization
because glucose-6-phosphate will not fit into the active site of enzyme
3.
Fructose-6-phosphate, This is simply a rearrangement called an isomerization
because glucose-6-phosphate will not fit into the active site of enzyme
3. -
Fructose-6-phosphate
 Fructose-1,6-diphosphate, This is a phosphorylation reaction. It is
also an endothermic reaction which requires energy of ATP.
Fructose-1,6-diphosphate, This is a phosphorylation reaction. It is
also an endothermic reaction which requires energy of ATP. -
Fructose-1,6-diphosphate
 DHAP, At this stage, there is cleavage producing two isomers. Isomers
have the same chemical makeup, but different structure.
DHAP, At this stage, there is cleavage producing two isomers. Isomers
have the same chemical makeup, but different structure. -
DHAP
 PGAL,
Here, DHAP is converted to PGAL by enzyme 6. There is an isomerization
reaction.
PGAL,
Here, DHAP is converted to PGAL by enzyme 6. There is an isomerization
reaction. -
2PGAL
 1,3-diphosphoglycerate, This is a phosphorylation. Phosphate has come
from H3PO4 rather than
ATP. It is also oxidation. Two electrons carried by a hydrogen
atom are lost to NAD+ to from NADH.
1,3-diphosphoglycerate, This is a phosphorylation. Phosphate has come
from H3PO4 rather than
ATP. It is also oxidation. Two electrons carried by a hydrogen
atom are lost to NAD+ to from NADH. -
1,3-diphosphoglycerate
 3-phosphogycerate, This is a dephosphorylation, phosphate is accepted by ADP
to form ATP.
3-phosphogycerate, This is a dephosphorylation, phosphate is accepted by ADP
to form ATP. -
3-phosphogycerate
 2 phosphoglycerate, This is simply an isomerization.
2 phosphoglycerate, This is simply an isomerization. -
2 phosphoglycerate
 phosphoenolpyruvate, This releases water, therefore it is a dehydration
reaction.
phosphoenolpyruvate, This releases water, therefore it is a dehydration
reaction. -
phosphoenolpyruvate
 pyruvate, This is a dephosphorylation, the phosphate is donated to ADP to
form ATP. It is an exothermic reaction.
pyruvate, This is a dephosphorylation, the phosphate is donated to ADP to
form ATP. It is an exothermic reaction.
In the steps of glycolysis, steps 1-5 are the activation phase of glycolysis. It is the preparation for energy extraction. Steps 6-10 is the energy extraction. These steps happen two times for every glucose. The end product is a net of 2 ATP (because it takes 2 ATP to move into the mitochondria), and 2 NADH.
Oxidation of Pyruvate
This step is the middle step between glycolysis and the citric acid cycle. It uses pyruvate and produces Acetyl CoA. Acetyl CoA is later used in the citric acid cycle for energy extraction. This occurs in the mitochondria. The final product is 2 NADH.
Citric Acid Cycle
The Citric Acid Cycle is also called the Krebs cycle, named after its discoverer. It is the main pathway for respiration. It is called a cycle because the starting compound is returned after the series of reactions in complete. This process occurs in the mitochondria. The steps of the Citric Acid Cycle are as follows:
-
Acetyl CoA + oxaloacetate
 citrate, This is a hydration reaction. Acetyl CoA is recycled and
returns to oxidation of pyruvate.
citrate, This is a hydration reaction. Acetyl CoA is recycled and
returns to oxidation of pyruvate. -
Citrate
 isocitrate, Steps 2 and 3 are really together. This is an
isomerization with an intermediate cis-aconitate
isocitrate, Steps 2 and 3 are really together. This is an
isomerization with an intermediate cis-aconitate -
The same as above
-
Isocitrate
 oxalosuccinate, This is a redox reaction. Isocitrate is oxidized to
oxalosuccinate.
oxalosuccinate, This is a redox reaction. Isocitrate is oxidized to
oxalosuccinate. -
Oxalosuccinate

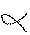 -ketoglutarate,
This is a decarboxylation because 6 carbons goes down to 5 carbons.
-ketoglutarate,
This is a decarboxylation because 6 carbons goes down to 5 carbons. -
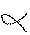 -ketoglutarate
-ketoglutarate  succinyl-CoA, This is similar to oxidation of pyruvate. It is a
decarboxylation, a redox reaction (the oxidation of
succinyl-CoA, This is similar to oxidation of pyruvate. It is a
decarboxylation, a redox reaction (the oxidation of
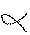 -ketoglutarate),
and a bonding of CoA to
-ketoglutarate),
and a bonding of CoA to
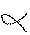 -ketoglutarate.
-ketoglutarate. -
Succinyl-CoA
 succinate, This is an
exothermic reaction. It releases energy which is used to form GTP (GTP
is more versatile than ATP), and it loses CoA.
succinate, This is an
exothermic reaction. It releases energy which is used to form GTP (GTP
is more versatile than ATP), and it loses CoA. -
Succinate
 fermarate, This is a redox
reaction where succinate is oxidized. FAD is used because there isn't
enough energy to reduce NAD+.
fermarate, This is a redox
reaction where succinate is oxidized. FAD is used because there isn't
enough energy to reduce NAD+. -
Fermarate
 malate, Simply a
hydration.
malate, Simply a
hydration. -
Malate
 Oxaloacetate, This is back to
what we started with. It is a redox reaction where malate is oxidized.
Oxaloacetate, This is back to
what we started with. It is a redox reaction where malate is oxidized.
This cycle happens twice for every one glucose molecule. The end results of the citric acid cycle is 2 ATP, 6 NADH, and 2 FADH2. The NADH and FADH2 move onto the Electron Transport Chain. This process is aerobic, but there is no direct input of O2. The oxygen is used on the Electron Transport Chain (ETC), and in the process, recycles NAD+ and FAD. If this didn't occur, citric acid cycle could not proceed. Each successive step in the cycle results in a decrease in energy stored in the molecule's bonds.
Electron Transport Chain
Most of the energy obtained from glycolysis and the citric acid cycle is stored in the high energy elections of FADH2 and NADH. This energy is not directly useful, so the electron transport chain (ETC) is used to convert this energy into useful ATP. The ETC is made up of three protein complexes: NADH dehydrogenase, cytochrome b-c, and cytochrome oxidase. There are also two carrier molecules in the ETC: coenzyme Q and cytochrome C. A diagram of the ETC is helpful.
In the ETC, each NADH produces 3 ATP, and each FADH2 produces 2 ATP. Each NADH from glycolysis is trapped outside the mitochondria, so it passes its electrons through the mitochondria membranes. These NADH produce only 2 ATP. Electrons are transferred down the ETC according to electronegativity.
Click to see a summary of Cellular Respiration. A total of 36 ATP is ideally generated.